Method diagram
Method and installation description
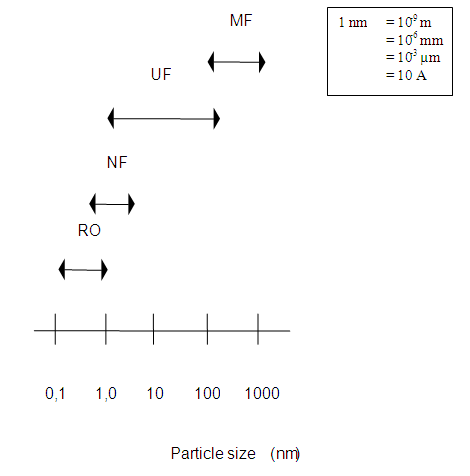
Reverse osmosis (or RO in short) is a pressure-driven membrane process with a separation range between 0.1 and 1 nm. Thus reverse osmosis membranes have high retention for bacteria, viruses and micro particles. Bivalent and some univalent ions are also blocked by the membrane.
An osmotic pressure difference is created in the membrane because reverse osmosis membranes are able to realise high retentions. A high salt concentration forms on the supply side, and a low salt concentration on the other side of the membrane (permeate side). Natural forces try to create a thermodynamic balance between both fluids separated by the membrane. In other words, the water with a low ion content diffuses through the membrane and decreases the concentration of ions on the other side. This process is called osmosis. There will then be a height difference between the two liquids. The height difference is related to a pressure - Osmotic pressure.
If pressure, which is higher than the osmotic pressure, is applied to the highly concentrated ion water, the clean water is forced to diffuse through the membrane to the side with a low ion concentration. This process is referred to as reverse osmosis. The level of this pressure is determined by the concentration of ions in the supply flow.
A reverse osmosis membrane consists of spiral-wound polyamide membrane layers (see figure below). At the edge of the membrane, the wound layers are sealed using a cap. A permeate collection tube is located in the centre of the wound module. All the clean water is passed through the spiral windings and collects in this tube.
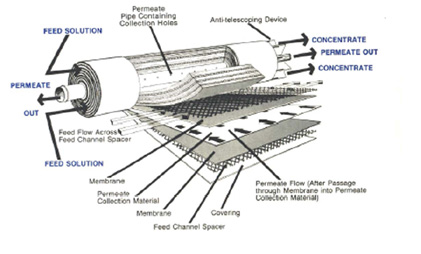
A full-scale installation is comparable with nano-filtration, in typical cross-flow set-up. The installation consists of one or multiple spiral-wound RO membranes in series or parallel. The series configuration provides a purer permeate but results in a higher amount of concentrate. A parallel configuration produces a lower quality permeate than the series configuration, but makes maximum use of the concentrate to minimise this volume.
Specific advantages and disadvantages
The specific advantages and disadvantages of reverse osmosis have been listed below:
Advantages:
- Reduction salt content and dissolved matter content in brackish water;
- Reduction in heavy metals;
- Reduction in nitrates and sulphates;
- Reduction in colour, tannins and turbidity;
- Softens hard water;
- chemical-free - e.g. needs no salt or chemicals during operation;
- High retention for salts and particular univalent ions (up to >99%);
- Disinfection, including viruses.
Disadvantages:
- Higher operating costs;
- High energy costs;
- Higher discharge volumes, higher concentrate volume than NF;
- High operating pressure than NF;
- Requires supply water to be treated (pre-filtration 0.1 - 20 microns);
- Reverse osmosis normally provides water with aggressive pH level (in other words, a low or high pH in water with few ions);
- Membranes sensitive to free chlorine.
Application
The main use of reverse osmosis is salt removal in brackish water and sea water, in preparation for drinking water. Large-scale versions of this technique are particularly popular in the Middle East. In the industrial sector, osmosis is used for the production of process water and high quality boiler supply water.
The technique has been adopted from wastewater purification, and is also used for manure processing. In manure processing, reverse osmosis is primarily used as the final purification step in thin manure fraction or in condensate derived from evaporation or drying. Membrane techniques for separating thin manure (and manure condensate) are now so well developed that small-scale implementation is also possible.
Boundary conditions
Reverse osmosis has stricter requirements for supply water. The quality of supply water must comply with the requirements of the membrane supplier, according to the accompanying technical information sheet. Here are a few examples:
- Maximum 0.5 ppm Fe/Al/Zn/Mn;
- SDI value of water for SDI15500 < 5;
- No free chlorine present in the supply water (resistance only 1000 ppm hours fee Chlorine);
- Maximum water temperature of 40 – 50 °C
- Maximum operating pressure 45 bar;
- pH range 3- 10, during cleaning pH transitory 2-12 (always consult the technical notes or the supplier).
Effectiveness
RO can be implemented for removing the following parameters (removal yield indicated in brackets):
- Dissolved matter (>95%);
- Harmful micro-organisms, e.g. bacteria, protozoa, algae, fungi (>99%);
- Persistent organic matter (75-99%);
- Organic compounds (>99%);
- Nutrients (incl. phosphates);
- Metals (>90%).
- Inorganic salts (e.g. sulphates).
Environmental issues
In reverse osmosis, just about all ions are concentrated into a watery flow – the retentate. This flow is a thickened version of the supply flow. Thickening is determined by the desired permeate quality, the design of the installation and the retention of the membrane.
The concentrated flow must be discharged if it cannot be put to good use in the process. When discharging, the discharge norms in the permit must be compared with the quality of the concentrate. An alternative may be to evaporate the concentrate.
Costs
The cost price for a typical reverse osmosis installation of 40 m³/day, with the process parameters below, lies between € 15.000 and € 20.000. This cost excludes on-site installation. The process characteristics for the case above are:
- Recovery 75 %;
- CIP tank;
- Average energy consumption 2.2 kW;
- 6 RO membranes;
- Configuration;
- Operating pressure 12 bar;
- Maximum recovery 75%;
- Nominal retention 90 - >99%;
- Working temperature 13-30°C;
- inflow pressure pump 3 -6 bar;
- design temperature 15°C.
Comments
Very high pressures (20-40 bar) are used in RO. Necessary safety measures must be taken (e.g. safety on the machine) to limit the risk of working with high pressures.
Complexity
/
Level of automation
More than 95% of an RO installation can be automated. For example, the cleaning process can be fully automated. However, manual intervention is still needed to replace membranes.
References
- Bonnélye V., Guey L., Del Castillo J., Desalination, Volume 222, Issues 1-3, 1 March 2008, Pages 59-65, 2008
- EIPPCB, Reference Document on BAT in Common Waste Water and Waste Gas Treatment / Management Systems in the Chemical Sector, draft February 2009 (revision upon release)
- Handbook on Wastewater. Dutch Membrane Guide, version 2.0, 1996
- http://www.kochmembrane.com/prod_spiral.html
- Mulder M., Basic Principles of Membrane Technology, Kluwer Academic Publishers, Dordrecht (NL), 1996.
- TNAV, supplier survey, 2008
- VITO-SCT, revision of technical notes WASS, 2008
Version February 2010

